 
In molecules with more than three atoms, there are many more possible geometries. The main geometries without lone pair electrons are: linear, trigonal, tetrahedral, trigonal bipyramidal, and octahedral. However, with a triatomic molecule (three atoms), there are two possible geometries: the atoms may lie on a line, producing a linear molecule, or not, producing a bent molecule. An example of the complexities which arise with polyatomic molecules is molecular geometry: how are the atoms in the molecule arranged with respect to one another? In a diatomic molecule, only a single molecular geometry is possible since the two atoms must lie on a line. Molecular geometry, on the other hand, depends on not only on the number of electron groups, but also on the number of lone pairs. The number of valence electrons in the central atom must be determined. Geometry molecular chart electron1 electron domain molecular geometry electron domain geometry part 1 Molecular geometry chartGeometry molecular electron vsepr molecule theory sampletemplates lone vs atoms predict xef2 bonded socratic steric trigonal electrons. It should be the atom with the highest electronegative potential. A polyatomic molecule contains more than two atoms. Chapter 5.1: predicting the geometry of molecules. PRACTICE PROBLEMS : Use the electron dot structure (Lewis structure) and the electron geometry table to determine the steric number and electron geometry. What is the steric number and electron geometry of PH5? What is the steric number and electron geometry of BrF3? Its Lewis structure consists of double bonds between the central carbon atom and each oxygen atom. Carbon dioxide is another example of a molecule which falls under the AB2 AB 2 category. If all the electron groups are bonded, with no lone pairs, then the electron geometry and molecular geometry are the same. The HBeH H Be H bond angle is 180o 180 o because of its linear geometry. The H-O bond length is 97 pm, while the O-Cl bond length is 169.3 pm respectively. The H-O-Cl bond angle decreases to about 103.3°. The lone pair-bond pair repulsions in HOCl distort the shape and geometry of the molecule. Arrange them in the way that minimizes repulsion (as far apart as possible). The ideal bond angle in a symmetrical tetrahedral molecule is 109.5°. 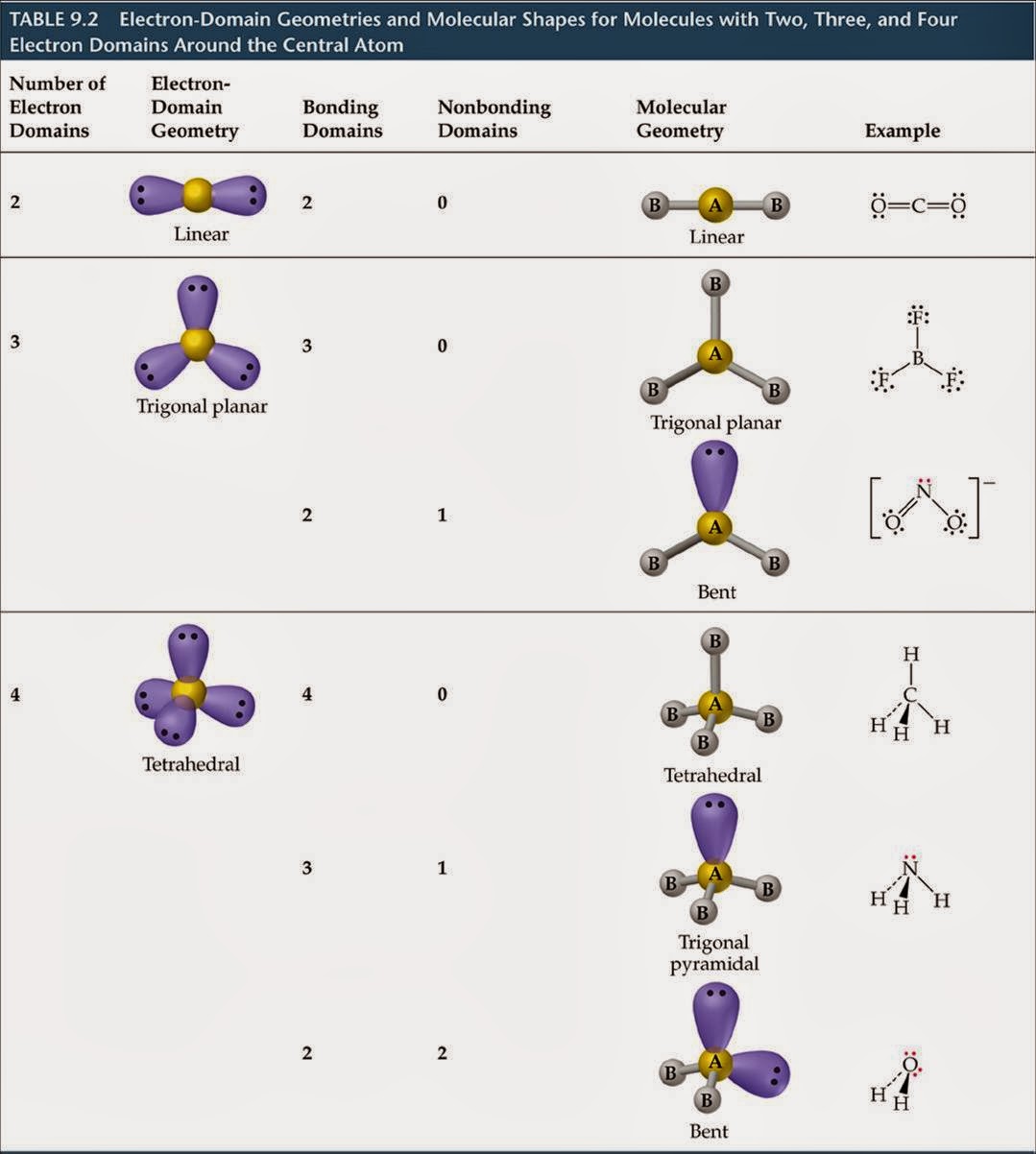
Count the number of atoms and lone pairs of electrons on the central atom (steric number) 3. If lone pairs of electrons, electrons not bonded to other atoms, are located in the molecule, this will change the molecular geometry, not the electron geometry. Draw the Lewis structure for the molecule. What is the steric number and electron geometry of SH2? Electron geometry is the arrangement of electron groups. You also treat double and triple bonds as one group instead of 3 just like you did in molecular shape. However, with electron geometry (steric number) lone pairs and bonds are treated the same. It is the 3D arrangement of all the atoms in a particular molecule. Molecular geometry, on the other hand, helps us understand the entire atom and its arrangement. Electron geometry teaches us about the arrangement of different electron groups. What is the steric number and electron geometry of SO2? In molecular shape (molecular geometry) you treat the electron’s lone pairs and the bonds as two different and separate things. To help you out, we are shedding light on electron geometry vs molecular geometry in this article. Refer to the electron geometry table below.Įxamples: Use the electron geometry table to determine the steric number and electron geometry of each molecule. This means you count up the lone pairs and number of atoms attached to the central atom into one number (the steric number) and from there determine electron geometry. 8. You also treat double and triple bonds as one group instead of 3 just like you did in molecular shape. Molecular geometry chartElectron molecular geometries examples pairs geometry domain nonbonding different atoms positions only some Electron molecular geometry: chemistryMolecular geometry chart. In molecular shape (molecular geometry) you treat the electron’s lone pairs and the bonds as two different and separate things. What is the difference between electron geometry (electron domains / electron clouds / steric number) and molecular shape (molecular geometry)?īoth are very similar, but the difference is in how we treat that electron’s lone pairs versus the bonds. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
